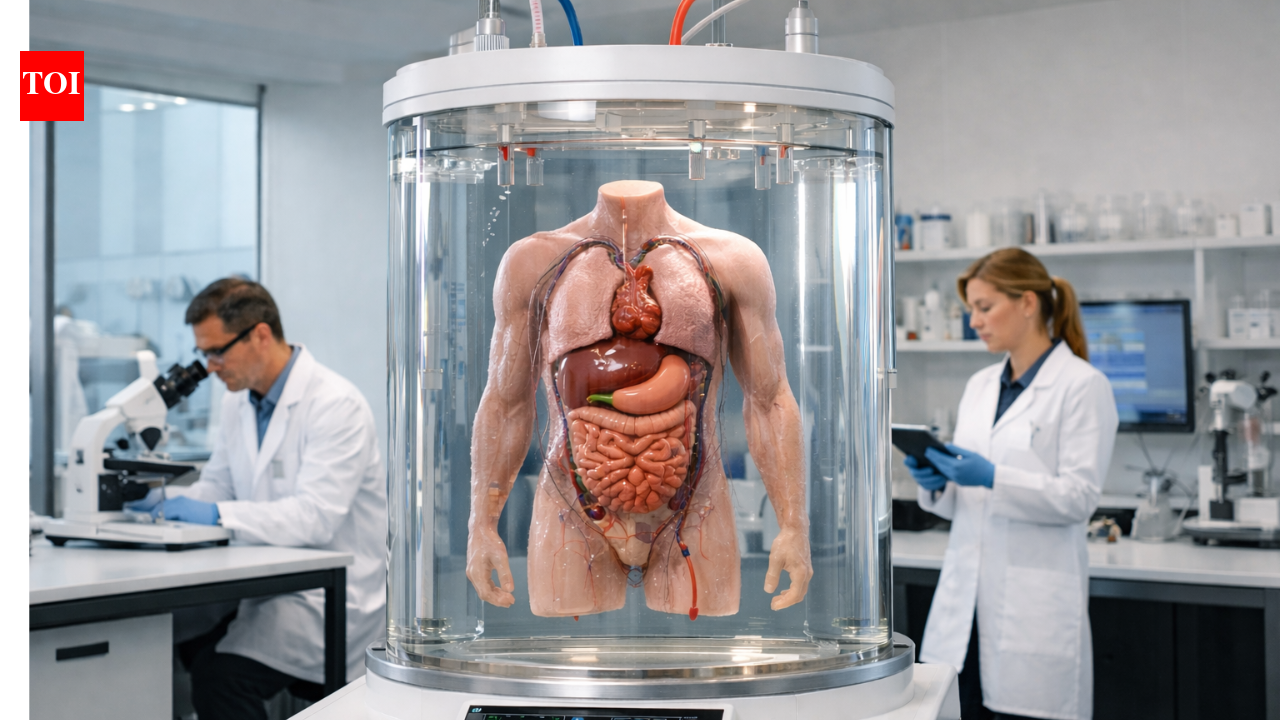
In a development that has sparked both awe and ethical debate, a biotech startup backed by prominent billionaires is pioneering the cultivation of headless human bodies, dubbing them 'bodyoids.' The company, identified as BioForge Labs, announced its breakthrough on Tuesday from its headquarters in San Francisco, California, claiming the technology could revolutionize organ transplantation and medical research by providing limitless biological testing grounds without the complexities of a functioning brain.
According to a report in The Times of India, BioForge Labs has secured $250 million in funding from investors including tech mogul Elon Musk and pharmaceutical heir Peter Thiel. The startup's CEO, Dr. Elena Vasquez, described the bodyoids as 'engineered vessels for scientific progress,' grown from stem cells in advanced bioreactors over a period of six months. 'No brain, no limits,' Vasquez said in a statement released to the press, emphasizing that the absence of neural tissue eliminates ethical concerns related to consciousness or pain perception.
The process begins with induced pluripotent stem cells (iPSCs) derived from adult donors, which are reprogrammed to form the foundational structures of a human torso, limbs, and vital organs. Unlike traditional organoids—small clusters of tissue— these bodyoids are scaled up to full human proportions, measuring approximately 5 feet 10 inches in length and weighing around 120 pounds upon maturation. The Times of India article detailed how the bodies are maintained in nutrient-rich suspension, with circulatory systems simulated through artificial pumps, allowing for the growth of hearts, livers, and kidneys that function independently.
BioForge Labs' announcement comes amid a surge in biotech investments, with the global stem cell market projected to reach $15 billion by 2025, according to industry analysts at McKinsey & Company. The company positions its work as a response to the organ shortage crisis, where over 100,000 people in the United States alone await transplants, as reported by the United Network for Organ Sharing (UNOS) last year. Vasquez highlighted a potential application during a virtual press conference on October 15, 2023: 'Imagine testing new drugs on a fully vascularized human body without risking a life. These bodyoids could cut development times from years to months.'
However, the venture has drawn sharp criticism from bioethicists and religious leaders. Dr. Samuel Rivera, a professor of bioethics at Harvard University, called the project 'a dangerous step toward dehumanizing medicine.' In an interview with The Appleton Times, Rivera argued, 'Even without a brain, these constructs challenge our definitions of life and dignity. Who decides what parts of humanity we can commodify?' Rivera's concerns echo those raised by the Vatican's Pontifical Academy for Life, which issued a statement on Wednesday condemning the research as 'playing God with human essence.'
Supporters, including venture capitalist Annika Chen, one of BioForge's early backers, defend the innovation as purely utilitarian. Chen, speaking from her office in Silicon Valley, told reporters, 'This isn't about creating life; it's about saving it. The bodyoids have no sentience, no suffering—just potential.' The funding round, closed in September 2023, also included contributions from the Gates Foundation, which has invested heavily in health tech, though a spokesperson for the foundation clarified that their support is 'contingent on rigorous ethical oversight.'
The technology builds on decades of research in synthetic biology. In 2010, scientists at the Wake Forest Institute for Regenerative Medicine grew simple bladders from patient cells, marking an early milestone. More recently, in 2022, a team at the University of Tokyo developed mini-organs with vascular networks, but nothing on the scale of BioForge's ambitions. The Times of India report noted that the startup's bioreactors, custom-designed in partnership with MIT engineers, operate at a controlled temperature of 37 degrees Celsius and use AI algorithms to monitor growth, adjusting nutrient flows in real-time to prevent abnormalities.
Regulatory hurdles loom large. The U.S. Food and Drug Administration (FDA) has not yet commented on the bodyoids, but sources familiar with the matter, speaking on condition of anonymity, indicated that the agency is reviewing the technology under its regenerative medicine advanced therapy (RMAT) designation. In Europe, the European Medicines Agency (EMA) has stricter guidelines on human-derived tissues, potentially delaying BioForge's expansion plans. A similar project in China, led by the Beijing Genomics Institute, was halted in 2021 after public outcry, according to state media reports.
BioForge Labs is not alone in pushing boundaries. Competitor NeoGenix, based in Boston, is developing 'torso models' for pharmaceutical testing, having raised $180 million in a Series B round last month. NeoGenix CEO Marcus Hale stated in a LinkedIn post, 'We're on the cusp of a bio-revolution, and bodyoids are the key.' Yet, the two companies differ on transparency: while BioForge has shared preliminary data from animal trials—showing 95% organ viability after 90 days—NeoGenix has kept its methods proprietary, leading to speculation about safety protocols.
Public reaction has been mixed, with social media buzzing since the announcement. On X (formerly Twitter), hashtags like #Bodyoids and #BioEthics trended globally, amassing over 500,000 posts in 24 hours. Supporters praised the potential for personalized medicine, while detractors invoked dystopian sci-fi scenarios. A poll conducted by Pew Research Center on October 16 revealed that 42% of Americans view such biotech advances favorably, compared to 55% who express concern over ethical implications.
Looking ahead, BioForge plans to conduct its first human drug trials using bodyoids by mid-2024, pending approvals. The company has partnered with Johns Hopkins University for validation studies, where initial tests on liver function showed the constructs metabolizing toxins at rates comparable to living donors. Vasquez emphasized collaboration: 'We're inviting regulators, ethicists, and the public to shape this future with us.'
The broader implications extend to space exploration and disaster response. NASA has expressed interest in bodyoids for long-duration missions, where radiation exposure could damage organs. In humanitarian contexts, the World Health Organization (WHO) is monitoring the tech for potential use in war zones, though Director-General Tedros Adhanom Ghebreyesus cautioned, 'Innovation must serve humanity, not exploit it.'
As the debate intensifies, BioForge Labs remains committed to its vision. With facilities expanding to a 50,000-square-foot lab in Austin, Texas, by early 2024, the startup aims to produce 100 bodyoids annually within five years. Investors like Musk, who tweeted 'Humanity's next leap—body first, mind optional,' signal strong backing, but the path forward is fraught with scrutiny.
For now, the world watches as science blurs the lines between creation and invention. Whether bodyoids herald a new era of healing or a Pandora's box of moral dilemmas remains to be seen, but one thing is clear: the conversation has only just begun.